Johan Widmark | 2025-08-29 08:00
Clinical validation de-risks regulatory pathway
A key achievement in Q2 was the first patient data from the whole blood study at St Thomas’ Hospital, confirming that Psyros delivers equivalent performance in whole blood compared to plasma. This not only supports the planned regulatory clinical performance study in 2025 but also de-risks the system’s clinical profile ahead of discussions with regulators and partners. In addition, a joint research project demonstrated rapid transfer of a cMyC biomarker assay to the Psyros platform, underlining the system’s broader potential beyond troponin and its versatility as a multi-analyte diagnostic tool.
Financing secured and board strengthened
Prolight’s SEK 100m (SEK 82.6m net after fees and set-offs) rights issue was fully subscribed, with strong participation from existing shareholders, management, and ITL, the company’s instrument manufacturing partner. The outcome eliminates near-term financing risk, securing the resources needed to complete development, validate pilot production, and prepare for the regulatory performance study without financial pressure. The election of Fredrik Alpsten as Chairman brings extensive international diagnostics experience and adds strategic weight as Prolight advances commercial negotiations.
Building traction for commercial launch
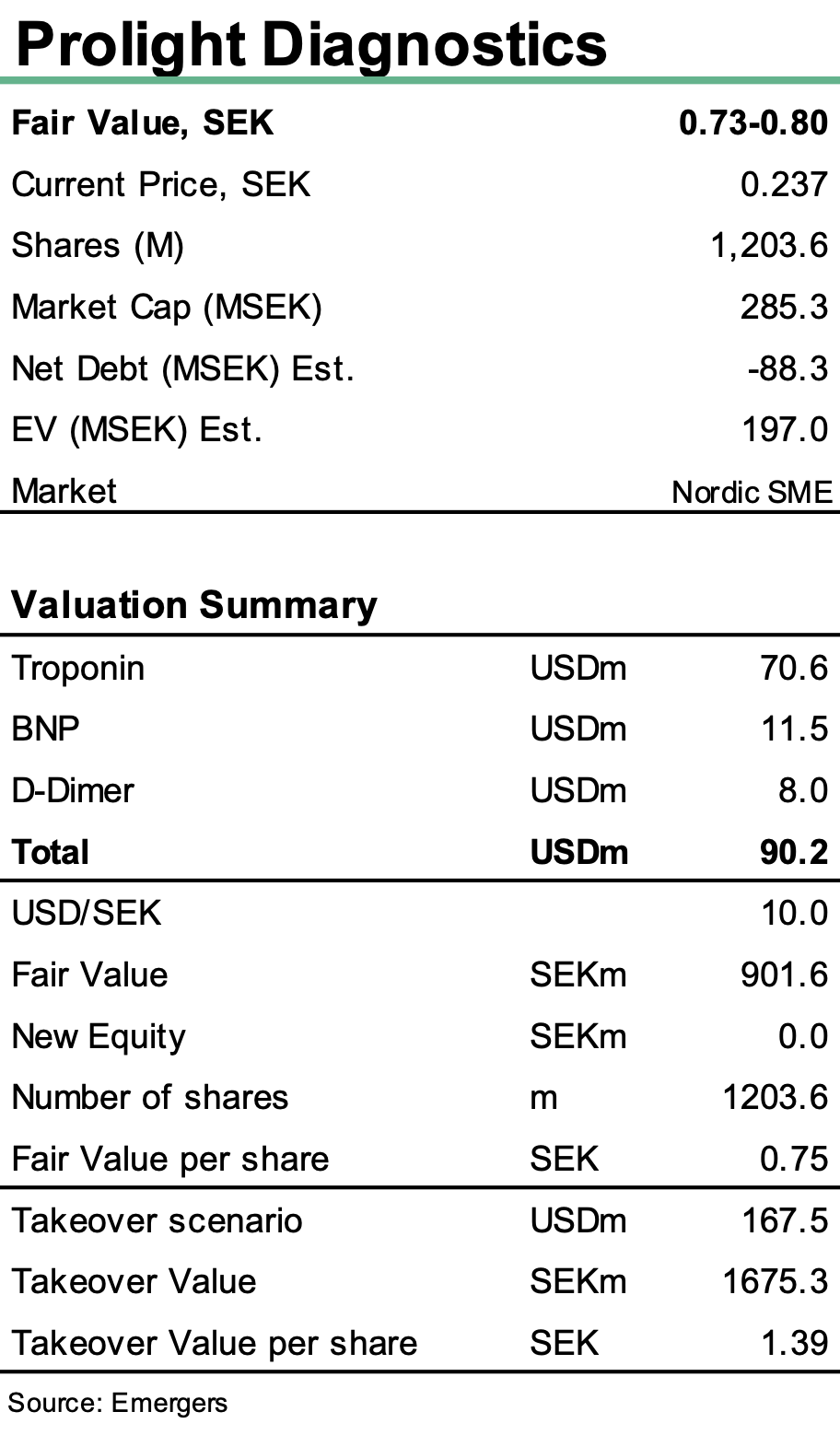
The company showcased its fully operational commercial prototype at the international congress ADLM 2025 in Chicago, attracting strong international attention and reinforcing market timing after positive biobank and whole blood results. Together with a strengthened IP position in Europe and Japan, this boosts Prolight’s credibility as it intensifies global partner talks. With financing secured and clinical risk reduced, we reiterate our fair value range of SEK 0.73–0.80 per share, with upside to SEK 1.35 in a successful partnering scenario. We expect commercial discussions to accelerate in the coming quarters, positioning Prolight for a transformative agreement.